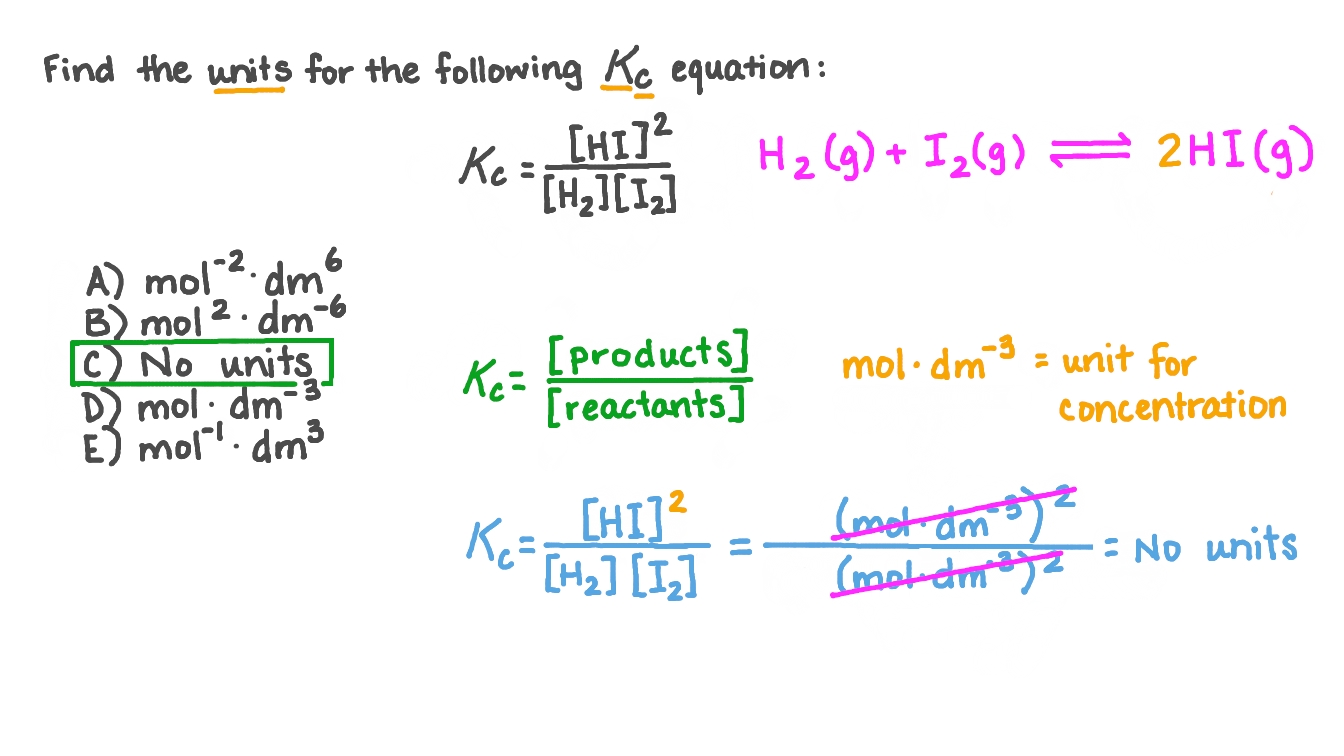
2 moles of an ideal gas at temp 27 degree c is heated isoermall from volume v to 4v .if R=2 cal/mol then the heat input in the process is approximately
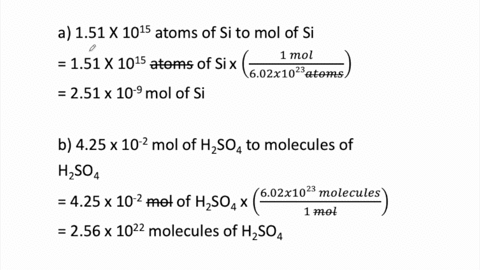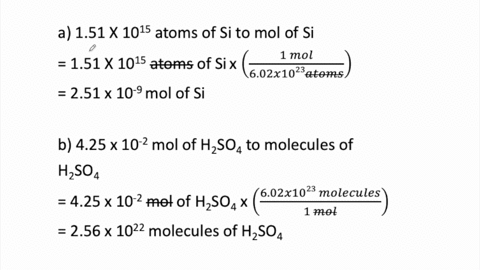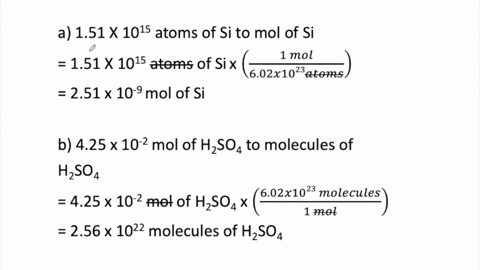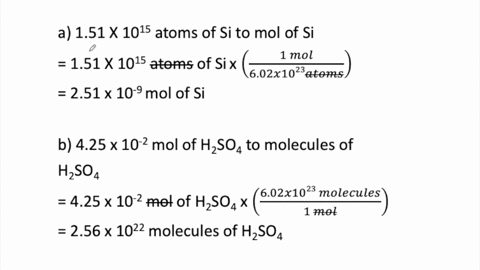
SOLVED:Perform the following conversions. a. 1.51 ×10^15 atoms of Si to mol of Si b. 4.25 ×10^-2 mol of H2 SO4 to molecules of H2 SO4 c. 8.95 ×10^25 molecules of CCl4

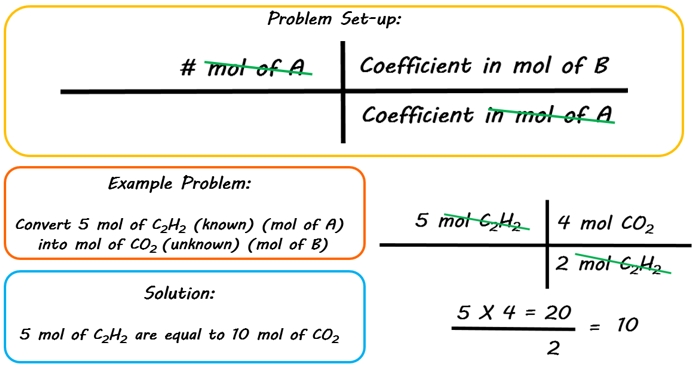
How to Convert from Moles of One Substance to Moles of Another Substance (Stoichiometry part 1) - YouTube

Two moles of PCl5 were introduced in a 2l flask and heated at 600K at equilibrium PCl5 was found to be 40
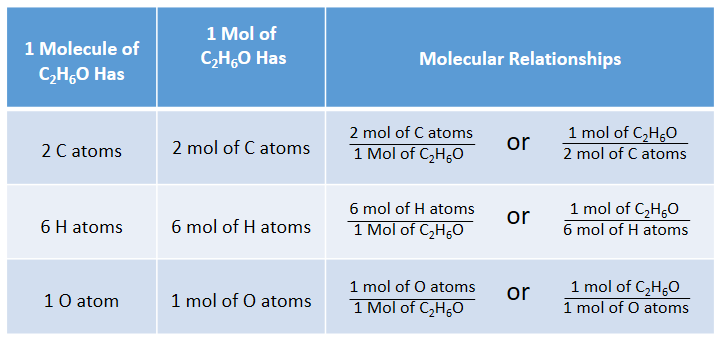
Moles and Molar Mass To find the molar mass of a compound: Example: Find the molar mass of sodium carbonate, Na 2CO3. Na 2 x 23.

The rate constant of a reaction is 9.2 × 10 5 mol 2 L 2 s 1. The order of the reaction is:A. 3B. 2C. ZeroD. 1
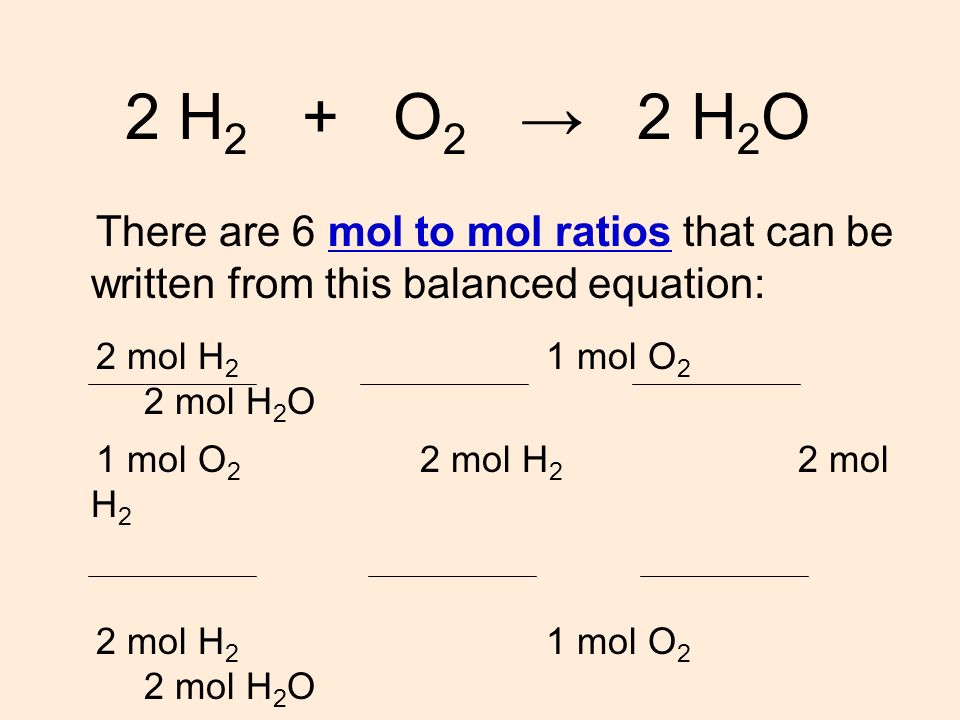
Mol ratio: coefficients of a balanced equation 2 H 2 + O 2 → 2 H 2 O 2 mol H 2 for every 1 mol O 2 In chemical calculations, mol ratios convert moles of. - ppt download